
Dental Admission
Test (DAT)
2024 Candidate Guide
©2024 American Dental Association. All rights reserved. | 800.232.1694 | ADA.org/DAT

IMPORTANT NOTE: COVID-19 may continue to have an impact on examination programs implemented by DTS. Candidates should
regularly monitor
ADA.org/DAT
, to understand the potential impact of COVID-19 on Examination Program administrations. Candidates
must comply with all required test center procedures, including those pertaining to COVID-19.
You are required to read this Guide before you apply to take the examination.
At the time of application, you will be required to confirm that you have read this Guide, understood its contents, and agree to
the policies and procedures contained herein.
Changes to the Dental Admission Test (DAT®) Program may occur after publication of this Guide. Updated PDFs will be posted to
ADA.org/DAT
. You will be subject to the policies and procedures currently in effect at the time of your test administration.
The Department of Testing Services (DTS) is a shared service of the American Dental Association (ADA) that
employs testing professionals and provides professional examination services in all relevant areas (e.g., test
design, content development, administration, analysis, and reporting). At the direction of the governing bodies
it supports, DTS advises on policy based on testing industry best practices and professional experience, and
implements policy for the orderly, secure, and fair administration of examination programs within its charge.

Dental Admission Test (DAT)
2024 Candidate Guide
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 1
Table of Contents
OVERVIEW ............................................................. 2
About this Guide and Examination Program ...... 2
Purpose of the Examination ............................... 2
Examination Fairness ......................................... 2
Ethical Conduct .................................................. 3
EXAMINATION CONTENT AND
PREPARATION MATERIALS ................................ 4
Examination Specifications ................................ 4
Scope of the Examination .................................. 4
Confidentiality of Examination Materials ............ 8
Examination Preparation .................................... 9
Practice Test ....................................................... 9
Test Drive ........................................................... 9
RESULTS INFORMATION ...................................10
Scoring of Examination ....................................10
Results Audits ...................................................10
Results Reports ................................................10
ELIGIBILITY REQUIREMENTS ............................12
FEES, APPLICATION AND TEST
ADMINISTRATION ................................................13
Examination Fees .............................................13
Extension Request Guide Notice .....................13
Rescheduling Fees ...........................................14
Partial Fee Waiver ............................................14
Retesting Policy ................................................15
Partial Testing ...................................................15
Administration Schedule ...................................16
Application Procedures .................................... 17
Test Center Procedures .................................. 18
Identification Policy ................................
.......... 18
Reschedule or Cancel a Testing
Appointment ..................................................... 19
No-Show Policy ............................................... 19
Emergencies on the Day of a
Testing Appointment ........................................ 20
Testing Problems on the Day of the
Testing Appointment ........................................ 20
Testing Accommodations ................................ 21
Unacceptable Forms of Documentation .......... 22
E
XAMINATION REGULATIONS
AND RULES OF CONDUCT ................................ 23
Rules of Conduct ............................................. 23
Privacy and Security ........................................ 24
Examination Misconduct .................................. 25
Irregularities and Appeals ................................ 28
Arbitration Requirement and
Agreement to Arbitrate .................................... 29

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 2
OVERVIEW
ABOUT THIS GUIDE AND EXAMINATION PROGRAM
This document is the official candidate guide to policies and procedures for the Dental Admission Test (“DAT
®
”
or the “Examination”). It provides information such as application and testing procedures, examination content,
the consequences of rules violations, and scoring. The governing body of the DAT is the American Dental
Association’s (ADA) Council on Dental Education and Licensure (“CDEL” or “Governing Body”). The DAT is
implemented by the Department of Testing Services (“DTS”), which is a shared service of the American Dental
Association. Examinations are administered by Prometric (“Prometric” or “Administration Vendor”). Collectively,
the Governing Body, DTS, and the set of activities, policies, and procedures occurring in support of this
examination are referred to as the “DAT Program” or simply the “Examination Program.”
The Council on Dental Education and Licensure (CDEL) would like to thank you for considering this examination.
CDEL appreciates the time and energy you are devoting in preparation. The agency takes its responsibilities
extremely seriously, and works diligently to help ensure examinations provide valid, reliable and fair evaluations
of candidate knowledge, skills, and abilities.
PURPOSE OF THE EXAMINATION
The DAT is an admission test designed to provide dental education programs with a means to assess an
applicant’s readiness and potential for success in these programs. Programs use the DAT in conjunction with
other admission tools that provide insight into candidate qualifications as they relate to core program
requirements.
Test results are just one factor considered in evaluating applicant potential. Validity studies conducted by DTS
have consistently shown that test scores in conjunction with academic performance are useful and effective in
predicting success in healthcare educational programs. The relative importance of each factor in the admission
process is determined by each educational program.
EXAMINATION FAIRNESS
Fairness, diversity, and inclusion are values that are of critical importance to society and to health professions.
The Governing Body and DTS have devoted and continue to devote substantial time and energy to these
considerations, to comprehensively consider the relevant issues and implement examination programs that are
fair, valid, and reliable, providing candidates with the opportunity to demonstrate their knowledge, skills, and
abilities in support of accurate and valid skill measurement. Fairness efforts are rooted in professional standards
as promulgated in the Standards for Educational and Psychological Testing
1
. This document—published by the
American Educational Research Association, American Psychological Association, and National Council on
Measurement in Education—provides professional guidance on all aspects of testing, and specifically notes
that fairness is fundamental to validity.
1
American Educational Research Association, American Psychological Association, National Council on Measurement in Education. (2014). Standards for Educational and
Psychological Testing.Washington, DC: Author.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 3
Fairness considerations are embedded throughout this Examination Program, affecting every aspect of how this
examination is constructed, administered, scored, and reported, appropriately recognizing the critical importance
of fairness to society and reflecting the core values of those who work closely and care deeply about this
program.
For additional information concerning how fairness considerations have been incorporated into this Examination
Program, please see the document entitled “The DAT and ADAT Programs: Overview of Policies and Procedures
Supporting and Promoting Fairness” on the Examination Program website.
ETHICAL CONDUCT
Health care professionals play an important role in society by providing services that contribute to the overall
health and well-being of individuals and their communities. In light of this responsibility, such professionals must
behave ethically at all times. This obligation begins at the time of application to school and continues through the
educational process, the licensure process, and the entirety of professional practice.
Members of the dental profession voluntarily abide by the ADA Principles of Ethics and Code of Professional
Conduct in the interest of protecting patients and maintaining the trust of society. The ADA Principles of Ethics
Conduct can be found at ADA.org/ethics.
Applicants are expected to abide by these ethical standards and to read, understand, and comply with the
examination regulations and rules of conduct for this examination. The obligation to abide by these ethical
standards includes a commitment to honesty, truthfulness, full disclosure, accuracy, fairness, and integrity in all
matters pertaining to examinations completed now and, in the future (examination applications, examination
procedures, applications for licensure, etc.). Misconduct resulting in an irregularity in the examination process is a
very serious matter. Violation of the rules of conduct or examination regulations may result in civil liability, voiding
of examination results, retest penalties, or other appropriate penalties.
The Examination Program Governing Body, licensure boards, and the profession expect strong ethical behavior
from all candidates. The Governing Body annually publishes policies and procedures applicable to misconduct
and irregularities in the application and examination process. This information is available in later sections of this
document. The Governing Body expects all candidates to carefully read and understand this information and their
obligations as candidates for this examination.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 4
EXAMINATION CONTENT AND PREPARATION MATERIALS
EXAMINATION SPECIFICATIONS
The DAT
®
is composed of multiple-choice test questions (items) presented in the English language. It consists of
four sections: Survey of the Natural Sciences, Perceptual Ability, Reading Comprehension, and Quantitative
Reasoning. Both the U.S. customary system (Imperial System) and the metric system (International System) of
measurement are used. Additional information on test content is provided below.
The Examination Program Governing Body, state boards, and the profession expect strong ethical behavior from
all candidates aspiring to be dentists. The Governing Body annually publishes policies and procedures applicable
to misconduct and irregularities in the application and examination process. This information is available in later
sections of this document. The Governing Body expects all candidates to carefully read and understand this
information, recognizing and respecting the obligations held by each candidate in taking this examination.
SCOPE OF THE EXAMINATION
SURVEY OF THE NATURAL SCIENCES (100 ITEMS)
The Survey of Natural Sciences test section consists of 100 items from Biology, General Chemistry, and Organic
Chemistry.
Biology (40 items)
• Cell and Molecular Biology: cell metabolism (including photosynthesis, enzymology), cellular processes
(including membrane transport, signal transduction), thermodynamics, mitosis/meiosis, cell structure and
function, experimental cell biology, biomolecules, and integrated relationships
• Diversity of Life: viruses, Archaebacteria, Eubacteria, Fungi, Protista, Plantae, Animalia, and integrated
relationships
• Structure and Function of Systems: integumentary, skeletal, muscular, circulatory, lymphatic/immune,
digestive, respiratory, urinary, nervous/sensory, endocrine, reproductive, and integrated relationships
• Genetics: molecular genetics, human genetics, classical genetics, chromosomal genetics, genetic
technology, developmental mechanisms, genomics, gene expression, epigenetics, and integrated
relationships
• Evolution and Ecology: natural selection, population genetics/speciation, animal behavior, ecology
(population, community, and ecosystem ecology), and integrated relationships
General Chemistry (30 items)
• Stoichiometry and General Concepts: percent composition, empirical formulae, balancing equations,
moles and molecular formulas, molar mass, density, and calculations from balanced equations
• Gases: kinetic molecular theory of gases, Dalton’s, Boyle’s, Charles’s, and ideal gas law
• Liquids and Solids: intermolecular forces, phase changes, vapor pressure, structures, polarity, and
properties
• Solutions: polarity, properties (colligative, noncolligative), forces, and concentration calculations
• Acids and Bases: pH, strength, Brønsted-Lowry reactions, and calculations
• Chemical Equilibria: molecular, acid/base, precipitation, calculations, and Le Chatelier’s principle
• Thermodynamics and Thermochemistry: laws of thermodynamics, Hess’s law, spontaneity, enthalpies
and entropies, and heat transfer
• Chemical Kinetics: rate laws, activation energy, and half-life
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 5
• Oxidation-Reduction Reactions: balancing equations, determination of oxidation numbers,
electrochemical calculations, and electrochemical concepts and terminology
• Atomic and Molecular Structure: electron configuration, orbital types, Lewis-Dot diagrams, atomic theory,
quantum theory, molecular geometry, bond types, and sub-atomic particles
• Periodic Properties: representative elements, transition elements, periodic trends, and descriptive
chemistry
• Nuclear Reactions: balancing equations, binding energy, decay processes, particles, and terminology
• Laboratory: basic techniques, equipment, error analysis, safety, and data analysis
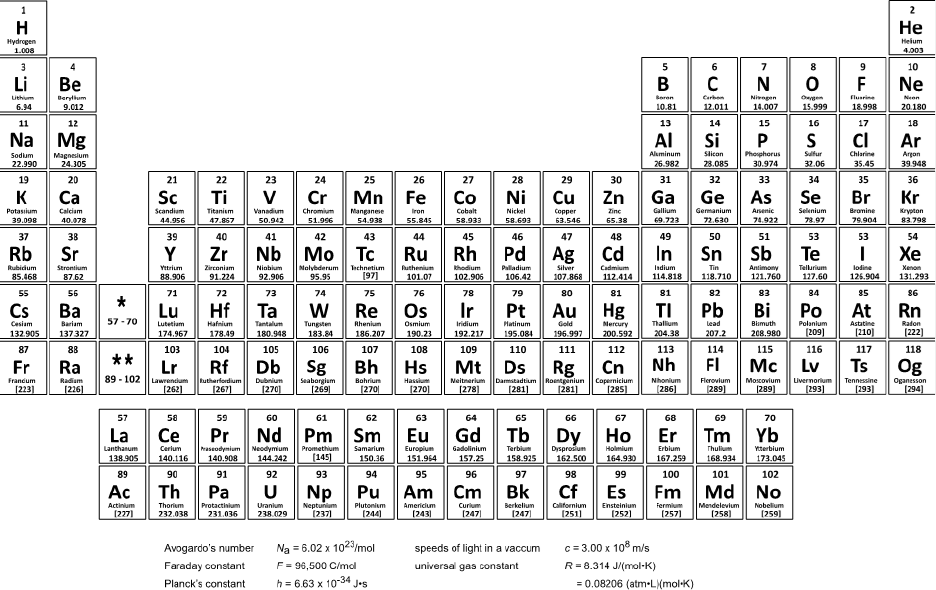
Note: An image of the periodic table of the elements is available to candidates as they complete the
General Chemistry section of the DAT:

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 6
Organic Chemistry (30 items)
• Mechanisms: Energetics and Structure – elimination, addition, free radical, substitution mechanisms,
and other mechanisms and reactions
• Chemical and Physical Properties of Molecules: Spectroscopy (1H NMR, 13C NMR, infrared, and multi-
spectra), structure (polarity, intermolecular forces (solubility, melting/ boiling point, etc.)), and laboratory
theory and techniques (TLC, separations, etc.)
• Stereochemistry (structure evaluation): Chirality, isomer relationships, and conformations
• Nomenclature: IUPAC rules and functional groups in molecules
• Individual Reactions of the Major Functional Groups and Combinations of Reactions to Synthesize
Compounds: Alkene/alkyne, aromatic, substitution/elimination, aldehyde/ketone, carboxylic acids and
derivatives, and other. For each area listed above, the following sub-areas apply: general, one-step,
and multi-step
• Acid Base Chemistry: Ranking acidity/basicity (structure analysis and pH/pKa data analysis), and
prediction of products and equilibria
• Aromatics and Bonding: Concept of aromaticity, resonance, atomic/molecular orbitals, hybridization,
and bond angles/lengths
Note on Mechanisms: The following examples show how curved arrows are used to represent organic chemistry
mechanisms on the DAT.
Double/full-headed curved arrows (A) represent the movement of electron pairs; single/half-headed (fishhook)
curved arrows (B) represent the movement of a single electron. Candidates will be expected to predict products
based on starting materials and curved arrows as well as predict curved arrows based on starting materials and
products.

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 7
Perceptual Ability (90 Items)
The Perceptual Ability Test is comprised of six subtests that assess the candidate’s ability to accurately perceive
object dimensions and mentally manipulate objects in space. This includes, for example, the ability to differentiate
among angles, or imagine how three-dimensional objects appear when viewed from different angles.
TOPIC DESCRIPTION
Apertures Evaluate a three-dimensional object and determine if it can pass through an opening.
View Recognition Visualize how an object would appear if viewed from different angles.
Angle Discrimination Rank a series of angles from smallest to largest.
Paper Folding Mentally unfold a piece of paper that has been folded one or more times and then hole-
punched.
Cube Counting Evaluate a stack of cubes and determine how much of each cube is exposed.
Spatial Relations / 3D
Form Development
Identify the three-dimensional shape that a flat pattern produces when folded
in a specific way.
Reading Comprehension (50 items)
The Reading Comprehension Test contains three reading passages on various scientific topics. Prior
understanding of the science topics is not a prerequisite to answering the test items. The reading passages
require the ability to read, comprehend, and thoroughly analyze basic scientific information.
Quantitative Reasoning (40 items)
• Mathematical Problems: Algebra (equations and expressions, inequalities, exponential notation, absolute
value, ratios and proportions, and graphical analysis); Data Analysis, Interpretation, and Sufficiency;
Quantitative Comparison; and Probability and Statistics

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 8
• Applied Mathematics (Word) Problems
At right is an image of the digital calculator available to
candidates for the Quantitative Reasoning section of this
examination:
All examination questions are reviewed annually by a team
of subject matter experts before they are used for
examination purposes. This process helps to ensure that
the questions reflect the most recent research and
guidelines. Changes to the test specifications for the DAT
may occur after the publication of this Guide. If changes
occur, a new Guide will be posted to ada.org/DAT and
listed in the document entitled Recent and Forthcoming
Updates to Examinations. This latter document is available
at ADA.org/testing.
CONFIDENTIALITY OF EXAMINATION MATERIALS
Examination items represent confidential, copyrighted intellectual property. Obtaining, using, or distributing
examination questions, also referred to as examination items, is strictly prohibited, regardless of the method
employed (memorization, recording, copying, or other means). This prohibition includes the discussion,
distribution, or online posting of remembered examination questions or answers, in whole or in part.
The use or sharing of examination items violates the examination regulations and rules of conduct of this testing
program. Such activities could provide an unfair advantage and threaten the validity and credibility of the
examination. Since all examinations are copyrighted property, these prohibited activities also violate federal
copyright laws.
The Department of Testing Services investigates all reports of candidates’ alleged production, misuse, or
sharing of current examination materials, and will pursue formal action against anyone who violates the
Examination Regulations or federal copyright law. Violations could result in the voiding of examination
results, legal action, or other appropriate penalties.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 9
EXAMINATION PREPARATION
The DAT Program recommends candidates use textbooks and lecture notes as primary sources for study.
A number of resources are used by subject matter experts to create questions for the DAT. These resources
include the following reference texts:
• Practice Test
• Reference Texts
• Prometric Online Tutorial
The DAT Program does not endorse any specific test preparation courses and has no data on the content or
efficacy of test preparation courses designed to prepare candidates for the DAT.
PRACTICE TEST
Candidates interested in preparing for the DAT may utilize practice questions available at ADA.org/dat. All
practice questions are copyrighted.
The intent of practice test questions is to help candidates understand the types of questions that will be
asked on the examination. Candidates are cautioned not to limit preparation for the examination to the
review of practice questions.
TEST DRIVE
Candidates can additionally become familiar with the test administration experience through Prometric’s Test
Drive on Prometric.com. This 30-minute overview includes the following experiences candidates will encounter at
the test center on their official day of testing:
• The scheduling and registration process
• The complete check-in process
• Introduction to test center staff and surroundings
• A 15-minute sample test (a generic test, not examination-specific) demonstrating the testing process
Visit Prometric’s Test Drive at prometric.com for further details and pricing.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 10
RESULTS INFORMATION
SCORING OF EXAMINATION
DAT
®
results are reported as scale scores. These scale scores are neither raw scores (i.e., the number of
questions answered correctly) nor percentiles. The calculation of scale scores is accomplished using
sophisticated psychometric equating procedures to accurately and fairly evaluate candidate skills. Using scale
scores it is possible to meaningfully compare the performance of candidates who have completed this
examination, even if candidates have completed examination forms containing different examination questions.
Candidates are not penalized for guessing.
Some questions on the test are experimental and are not scored. Data collected on unscored questions is used to
determine whether those questions pass psychometric standards and would be appropriate for use in future test
administrations. Unscored questions look the same to candidates as scored questions.
DAT
®
scores range from 1 to 30, and are reported in one-point increments. Each educational program makes its
own determination as to what constitutes an acceptable score. As such, there is not an official passing score for
this examination.
Candidates often ask whether they have achieved a good score. To interpret the results of your examination, the
Examination Program recommends consulting the most recent examination norms, which provide information on
candidate’s percentile standing on the examination. This information is available in the following report, which is
publicly available on the Examination Program website:
DAT User’s Manual
The above report is also available to educational programs as they interpret and use candidate test results for
admission purposes.
RESULTS AUDITS
As a routine part of quality assurance procedures, results are audited for accuracy before they are distributed.
Candidates can also request to have their examination results audited or checked for accuracy an additional time,
beyond that indicated above. To request this second audit, login to your account and follow the audit request
instructions. Additional fees will apply (see the Examination Fees section of this Guide). Results audits require
approximately four to six weeks to complete, and must be requested within 30 days of the reporting date indicated
on the official report of results.
RESULTS REPORTS
After a testing session has ended, an unofficial score report will be provided at the test center, unless the test is
taken over multiple days. Scale scores are reported on the unofficial score report. This report is a personal copy;
if an unofficial score report is not provided after completing the examination, candidates should contact the testing
program within five days via an email sent to datexam@ada.org.
The unofficial score report provided by the Administration Vendor is subject to audit as part of the Governing
Body’s quality review process. Falsification and/or misrepresentation of score reports is viewed as an egregious
violation of Examination Program policies, and could result in cancellation of scores, imposition of a mandatory
two-year wait period to retest, or the possibility of a lifetime ban from taking the examination. Such misconduct
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 11
may also have implications for the candidate’s ability to test on other examinations implemented by the ADA’s
Department of Testing Services.
Scores cannot be voided at the request of a candidate after completing any part of the examination.
At the time of application, candidates are asked to select schools and/or programs to receive official results. In so
doing, candidates grant DTS permission to release official results to these schools. Results will be released only
upon authorization, or by decision of the Examination Program in accordance with Examination Program policies
(e.g., in the case of irregularities or falsification of information). Official results are reported electronically within
three to four weeks of the testing date to the selected schools and will be posted to the candidate’s My Testing
Account page at ada.org/dentpin. If a candidate has tested more than once, a history of all testing attempts is
reported.
Candidates should confer with their advisors regarding test results. At the time of application, candidates can
request that scores be made available to advisors at no additional charge.
If a candidate requests on their application that scores be sent to any dental school, then the candidate’s scores
will also be sent to ADEA AADSAS. If a candidate requests to have their scores sent to any of the dental schools
in Texas, then their scores will also be sent to the Texas Medical & Dental Schools Application Service
[TMDSAS]. The testing program will report official scores to both schools and the aforementioned application
services within three to four weeks of the test date.
The examination application includes a list of potential results recipients. Schools selected at the time of
application are included in the examination fee, regardless of the number of schools selected.
A list of designated recipients cannot be edited or cancelled after it has been submitted. If no schools are
selected on the examination application, then permission has NOT been granted to release official
scores. Candidates must then submit score report requests separately.
Requests for additional score reports must be submitted using the score report request form available at
ADA.org/DAT. Additional fees apply when sending reports to schools or other recipients not selected at
the time of application (see the Testing Fees section of this Guide).
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 12
ELIGIBILITY REQUIREMENTS
The DAT
®
Program does not discriminate on the basis of race, ethnicity, religion, gender, age, disability,
sexual orientation, or marital status.
Successful participants in the DAT Program typically complete at least one year of college education, including
courses in biology, and general and organic chemistry. Advanced level biology is not required.
Most applicants complete two or more years of college before taking the test.
Dental school applicants are encouraged to take the DAT well in advance of the dental school admission cycle.
Applicants seeking admission to Canadian dental schools should confirm the acceptability of the DAT by
Canadian schools prior to applying for the test.

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 13
FEES, APPLICATION AND TEST ADMINISTRATION
EXAMINATION FEES
Fees are non-refundable and non-transferable. All fees are in US dollars. The following indicates 2024 testing
fees:
FEE TYPE DESCRIPTION FEE AMOUNT
Examination Fee This fee includes administration and official score reporting to schools
and programs selected at the time of application.
$540
Score Report Fee
(optional)
This fee covers score report requests made after the time of
application. There is no additional charge for score report requests
received at the time of application.
$50 per report
Score Audit Fee
(optional)
For a period of 30 days after a testing appointment, as an optional
service, the program is willing to audit a candidate’s results.
$65
Eligibility Extension
Fee
Candidates can extend their eligibility period for a fee. The extension is
for 45 days and available once per submitted application.
$135
EXTENSION REQUEST GUIDE NOTICE
Candidates may request a 45-day extension (weekends and holidays included) to their eligibility window by
logging into the Testing Account page.
Candidates may request only one extension per application and cannot have an examination appointment
currently scheduled. Any candidate with a scheduled appointment must cancel that appointment before
proceeding with an extension request; failure to do so will result in the extension request being denied. The
eligibility extension request does not supersede any other rules regarding a candidate’s eligibility.
There is an additional fee to obtain the 45-day extension. This fee must be paid at the time the extension request
is submitted and is non-refundable and non-transferable.
Please Note:
After DTS processes a candidate’s application, that candidate is eligible to test for a six-month period, unless
other considerations—including retest rules and program requirements—limit the eligibility period to a shorter
timeframe. Candidates are encouraged to consider their test administration date carefully, as any last-minute
requested changes (e.g., for medical reasons, personal reasons, or due to a missed testing appointment) may not
be approved. There is an additional fee to obtain the 45-day extension. This fee must be paid at the time the
extension request is submitted, and is non-refundable and non-transferable.

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 14
RESCHEDULING FEES
The fee to reschedule a testing appointment is determined by the amount of notice provided before the originally
scheduled administration date. Rescheduling fees are subject to change. Sales tax may apply to cancellation and
rescheduling fees. Base fees are as follows:
NUMBER OF DAYS PRIOR TO TESTING APPOINTMENT RESCHEDULING FEES
30 or more business days* $40
5-29 business days* $70
1-4 business days* $150
*Saturday and Sunday are NOT business days
PARTIAL FEE WAIVER
A limited number of partial fee waivers are available to candidates, in cases of severe financial hardship. The
waiver covers 50% of the exam fee, 50% of the exam fee for a candidate’s first retest, and 100% of the fee for a
full ADA DAT Practice Test. Fee waivers also cover any official score reports requested at the time of application.
The waiver does not apply to any charges associated with rescheduling or score reporting requested after the
time of initial application. Candidates who have previously received a fee waiver or who have already taken the
examination are not eligible. A portion of the available fee waivers are reserved for candidates from historically
underrepresented racial and ethnic groups (American Indian/Alaska Native, Black/African American, Latina/o, and
Native Hawaiian or Other Pacific Islander) who demonstrate financial hardship.
Candidates can obtain partial fee waiver forms from ADA.org/dat. The DAT Program will review all fee waiver
requests and make final decisions regarding fee waivers. Candidates must register for a DENTPIN
®
prior to
submitting a fee waiver request.
Candidates may qualify for a partial fee waiver if the following requirements are met:
• Demonstrated financial hardship
• First time taking the examination
• U.S. citizen or resident alien
• Received financial aid from the candidate’s educational institution
Required Documents:
• Fee waiver financial information form (ADA.org/dat)
• Educational institution financial aid award letter
For additional information on partial fee waivers please see the DAT Program website (ADA.org/dat).
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 15
RETESTING POLICY
Candidates are required to submit a new application and fee for each testing attempt. A testing attempt is defined
as any test administration where the candidate has been seated at a computer at a test center and electronically
agreed to the confidentiality statement to start the test.
• Candidates must wait 60 days between testing attempts on the DAT, with a maximum of four (4) such
administrations permitted during any 12-month period of time.
• Candidates with three (3) or more attempts on the DAT must apply for permission to test again, providing
proof of recent application to dental school with each subsequent application to test.
• Subsequent to the candidate’s fifth DAT attempt, the candidate may retest only once per 12-month
period.
• The preceding retest policies are not subject to appeal.
Candidates who are requesting additional testing beyond three attempts must submit their request in writing to
datexam@ada.org and must include one of the following acceptable forms of evidence of their demonstrated
attempt to apply to dental school within the previous 18 months:
• Copy of a completed and submitted ADEA AADSAS application
• Letter of rejection from a dental school
• Letter on school letterhead from a dental school admission officer encouraging the applicant to retest
• Letter on school letterhead from a college or university health profession advisor or instructor confirming
the applicant has applied to dental school
PARTIAL TESTING
Partial testing is not permitted. Applicants are required to take all four sections of the DAT. The lowest possible
scale score is reported for any assigned test not taken. Individuals unable to complete the DAT must submit a
new application and fee to retest.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 16
ADMINISTRATION SCHEDULE
The table below presents the DAT administration schedule. Candidates must report to the testing center at least
30 minutes prior to their scheduled appointment. The total administration time is five hours and 15 minutes,
including the optional tutorial, scheduled break, and survey.
DAT ADMINISTRATION SCHEDULE
Tutorial (optional) 15 minutes
Survey of Natural Sciences 90 minutes
Perceptual Ability Test 60 minutes
Scheduled Break (optional) 30 minutes
Reading Comprehension Test 60 minutes
Quantitative Reasoning Test 45 minutes
Post Test Survey (optional) 15 minutes
Total Time 5 hours 15 minutes
The optional break after the first two test sections is the only scheduled break. If a candidate chooses to take this
scheduled break, the testing session will resume automatically after 30 minutes have elapsed.
Taking a break at any other time is considered an unscheduled break. During an unscheduled break,
candidates may NOT access personal belongings or prohibited items, study, refer to notes or texts, use a
telephone, eat or drink any food or beverages, or leave the test center. Locker access during
unscheduled breaks is strictly prohibited.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 17
APPLICATION PROCEDURES
Before applying to take this examination, candidates must first obtain a Dental Personal Identification Number
(DENTPIN
®
). Candidates can register for a new DENTPIN or retrieve an existing DENTPIN at ADA.org/DENTPIN.
The DENTPIN
®
is a unique personal identifier used by U.S. dental and dental hygiene education system and
standardized testing programs, such as the Dental Admission Test (DAT), Advanced Dental Admission Test
(ADAT), and the Admission Test for Dental Hygiene (ATDH)—as well as application service programs such as the
American Dental Education Association (ADEA) Postdoctoral Application Support Service (ADEA PASS), the
ADEA Associated American Dental Schools Application Service (ADEA AADSAS), the Texas Medical & Dental
Schools Application Service (TMDSAS), the ADEA Centralized Application for Advanced Placement for
International Dentists (ADEA CAAPID), and the ADEA Dental Hygiene Centralized Application Service (ADEA
DHCAS). In each case, the DENTPIN
®
is used to uniquely identify individuals, and for the confidential and secure
reporting, transmission, and tracking of test scores and academic data.
Once a DENTPIN is obtained, candidates can submit an application through the Examination Program website. A
new application and fee must be submitted before each testing attempt. Application processing takes place
Monday through Friday during standard US business hours.
After DTS processes a candidate’s application, that candidate is eligible to test for a six-month period, unless
other considerationsꟷincluding retest rules and requirementsꟷlimit the eligibility period to a shorter timeframe.
Candidates are encouraged to choose their test administration date carefully, as any last-minute requested
changes (e.g., for medical or personal reasons) may not be approved.
When registering for a DENTPIN and submitting an application, all provided information must be accurate.
Candidates must use their legal name. When including a middle name, candidates must use either their full
middle name or a middle initial.
If the name on a candidate’s application fails to EXACTLY match the name appearing on IDs brought to the
Administration Vendor test center, the candidate will NOT be permitted to test. As a result, the testing
appointment and application fee will be forfeited and the candidate will be required to submit a new application
and fee before taking the examination.
Changes and corrections to the application (name, birth date, etc.) must be completed at least two weeks prior to
a scheduled testing appointment. Candidates are responsible for identifying any corrections or omissions and
must notify the testing program at dentpin@ada.org.
Updates made to contact information (address, email address, etc.) using the “Update Your DENTPIN
®
” page at
ADA.org/DENTPIN will NOT automatically update existing test applications and score report requests.
During the application and testing process, candidates will be required to provide their name, DENTPIN
®
address,
date of birth, and other requested information to allow proper identification by the testing program. This
information must be accurate. If it is determined that a candidate deliberately falsified personal information in the
DENTPIN
®
system, examination application, or at the test center, scores will be voided and all schools will be
notified. Possible repercussions associated with deliberate falsification include a required two-year waiting period
before taking the examination again, or a complete banning from the Examination Program and any other
examination program implemented by DTS.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 18
TEST CENTER PROCEDURES
The Administration Vendor will electronically capture the identity of each candidate biometrically (e.g., through
photograph, fingerprint or palm vein scan) before candidates can proceed with testing. Candidates must consent
to these procedures before they are permitted to test. Electronic capture of biometric data allows for a more
efficient return to testing after breaks. Biometric and other identifying information will be retained by the
Administration Vendor and will be utilized for identity verification at potential future test administrations (e.g.,
retesting).
Administration Vendor staff will visually inspect eyeglasses and hair accessories as part of check-in procedures.
Staff may also use an electronic detection wand to scan for electronic devices. Jewelry, except for wedding and
engagement rings, is prohibited. Updates to security protocols at check-in may change with little to no advance
warning. Candidates can view the current check-in procedures at the Administration Vendor’s website.
Administration Vendor staff will observe candidates at all times during the testing appointment. This observation
includes staff walking through the secure testing room, as well as video recording of the candidate’s examination
session. Administration Vendor staff are required to report behavior that might represent a violation of rules and
regulations.
Administration Vendor staff are not authorized to answer questions from candidates regarding examination
content, examination software, specific examination program policies, and scoring.
IDENTIFICATION POLICY
When you arrive at the administration vendor test center to take your examination, two original, current (not
expired) forms of identification (ID)one primary and one secondary will be required. An expired ID WILL NOT be
accepted, even if that ID is accompanied by temporary identification or documentation that a new, valid ID has
been requested.
The primary ID must be a government-issued ID with your photograph, name, and signature. Examples of
acceptable primary IDs include, but are not limited to a driver’s license, passport, or passport card. All IDs, with
the exception of passports, must be in English.
The secondary ID must contain your name and signature. Examples of secondary IDs include, but are not limited
to debit cards, library cards, or a credit card.
Only physical forms of IDs will be accepted by test center staff. Digital or paper copies of IDs will not be accepted.
WARNING! Your DENTPIN record (which is the name on your application) must match your IDs exactly or
you will be denied admission to testing and forfeit your testing/application fee.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 19
RESCHEDULE OR CANCEL A TESTING APPOINTMENT
To reschedule or cancel a testing appointment, candidates must either use the scheduling tools on the
Administration Vendor website or contact the Administration Vendor using the contact information provided on the
Administration Vendor website. This must be done in advance of the testing appointment. Additional fees apply
and must be paid directly to the Administration Vendor (see the Examination Fees section of this Guide). Local
test centers where candidates complete their examination cannot schedule, reschedule, or cancel your
appointment. Appointments must be canceled or rescheduled by the business day prior to the scheduled test, and
at least 24 hours in advance of the scheduled appointment. Candidates will receive a confirmation notice when
rescheduling their testing appointment; please retain a copy of this notice.
NO-SHOW POLICY
Candidates who do not appear for a scheduled testing appointment and do not cancel or reschedule their
appointment by the required time in advance of the test date will forfeit all testing fees. These candidates will be
required to submit a new application and provide corresponding payment to schedule a new appointment.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 20
EMERGENCIES ON THE DAY OF A TESTING APPOINTMENT
If an emergency occurs on the day of a testing appointment that prevents a candidate from sitting for their
examination, a written request for relief must be submitted to DTS. This request should include applicable
documentation and be sent to testingproblems@ada.org within five business days of the scheduled appointment.
Examples of emergencies and applicable documentation include, but are not limited to, the following:
• Sudden illness on the examination day. Provide a doctor’s note or hospital records confirming that you
were treated on the day of the examination.
• Death in the family on the examination day. Provide a copy of an obituary, prayer card, funeral service
program, or death certificate confirming that the relative passed away or services were held on the day of
the examination.
Testing appointments affected by emergencies occurring prior to the day of the scheduled appointment should be
handled through the Administration Vendor’s rescheduling and cancellation process indicated previously.
TESTING PROBLEMS ON THE DAY OF THE TESTING APPOINTMENT
If a candidate encounters a problem during their examination, the administration should not be resumed until the
issue has been documented and resolved by the test center administrator. If a candidate continues to have issues
with their testing experience after having requested such assistance, they should again alert test center staff and
request that staff resolve the issue. If the issue persists, the candidate should immediately discontinue testing.
Candidates who continue to test despite the presence of continued, significant issues waive their right to appeal
for a remedy on the basis of those encountered issues. Concerns not resolved at the time of testing must be
submitted in writing within five business days of the testing appointment to testingprobl[email protected]rg.
Candidates must contact testingprobl[email protected]g directly, and state the specific relief being requested. Upon
receipt of directly communicated information, DTS will conduct an investigation and notify the candidate of the
outcome. Candidates with documented, unresolved testing issues could be offered the courtesy of a retest within
30 days. If the candidate accepts the retest courtesy, the retest will replace the results of the initial test, and the
initial test results will be voided. Candidates who continue to test despite severe issues — and particularly those
who continue to test, wait for their results to be released, and call DTS afterwards upon receipt of a poor score —
are unlikely to obtain the remedy they seek.
Test center incident reports submitted on behalf of the candidate — and comments submitted by the candidate
via post examination surveys — are considered indirect communication to DTS. These indirect communications
may be considered by DTS as part of its general quality assurance procedures but would not result in specific
relief for the candidate.
Examination results cannot be canceled or adjusted under any circumstances.

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 21
TESTING ACCOMMODATIONS
The Examination Program provides reasonable and appropriate testing accommodations in accordance with the
Americans with Disabilities Act. These accommodations occur for individuals with documented disabilities or
medical conditions who demonstrate a need for accommodation, request an accommodation prior to testing, and
who are approved by the Examination Program to receive accommodations based on the information submitted.
Testing accommodations are offered to those with a qualified disability or a medical condition in order to offer
equal access to testing. Candidates must request testing accommodations with each application, but — for
subsequent administrations — will not be required to submit additional documentation covering the same
disability or condition.
Information concerning specific accommodations provided will not be shared outside of DTS, the test center, and
the Examination Program, and will not be indicated on examination results.
In considering a request from a candidate with a disability, the Examination Program is guided by a focus on
validity. Testing accommodations are provided so all candidates have the opportunity to demonstrate their
knowledge and skills, as opposed to having the measurement of their knowledge and skills inappropriately reflect
a disability.
To determine whether a candidate qualifies for accommodations under the Americans with Disabilities Actꟷor as
a result of a current medical conditionꟷthe Examination Program requires a complete evaluation of the candidate
as well as a completed and signed Testing Accommodations Request Form. A health care professional
appropriately qualified to evaluate the disability or medical condition must conduct the evaluation. For more
information on accommodations and an explanation of how to request testing accommodations, please consult
the Testing Accommodations Request area on the Examination Program website (see the section above entitled
“Application Procedures”).
If you have a documented disability recognized under the Americans with Disabilities Act and require testing
accommodations, you must submit the following three documents prior to testing:
1. A completed testing application
2. The Testing Accommodations Request Form through your DENTPIN account
3. Documentation to support the testing accommodation request
You may submit your testing accommodation request in one of the following ways.
Before applying to test
• You may submit your testing accommodation request prior to submitting your examination application.
The Examination Program will review your request, and if approved, the Examination Program will add
the approved testing accommodation to your record after you complete your examination application.
• To submit a testing accommodation request before applying to test, please sign into your DENTPIN
account and click “Submit Request” from the top ribbon menu. From the dropdown menu, select
“Accommodations Request”.
• Fill out and submit the Testing Accommodation Request Form. This will include a file uploader for
attaching the required supporting documentation. Requested testing accommodations should align with
the identified functional limitation, so that the adjustment to the testing procedure is compliant with federal
guidelines.

© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 22
OR
While applying to test
• During the application submission process, select “Yes” when asked “Are you requesting testing
accommodations under the Americans with Disabilities Act?”.
• Fill out the Testing Accommodation Request in the examination application, which will appear after the
payment page. This will include a file uploader for attaching supporting documentation. Requested testing
accommodations should align with the identified functional limitation, so that the adjustment to the testing
procedure is compliant with federal guidelines.
Candidates can schedule a testing appointment AFTER testing accommodation requests have been
approved. Testing accommodations cannot be added to a previously scheduled testing appointment. If
candidates schedule testing appointments before testing accommodations are approved, candidates will
be required to cancel the appointment and pay a rescheduling fee. Candidates requesting
accommodations must receive their eligibility email with approved accommodations before scheduling a
testing appointment.
UNACCEPTABLE FORMS OF DOCUMENTATION
Please do not submit the documents indicated below. They will not be accepted.
• Handwritten letters from health care professionals
• Handwritten patient records or notes from patient charts
• Diagnoses on prescription pads
• Self-evaluations
• Research articles
• Original documents (submit copies only)
• Previous correspondence to the Examination Program (DTS maintains copies of all correspondence)
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 23
EXAMINATION REGULATIONS AND RULES OF CONDUCT
RULES OF CONDUCT
Rules have been established that govern the administration of this examination to ensure results accurately
reflect candidates’ skills. Examination regulations and rules of conduct help preserve the integrity of the
examination process and provide standardized examination administration conditions that yield valid and reliable
results.
The Examination Program bears no responsibility for inaccurate information or inappropriate permissions received
from test center administrators. It is your personal responsibility to understand and comply with the Examination
Regulations indicated in this guide. If a candidate’s conduct is determined to violate the terms set forth in this
Guide, the Examination Program will act to strictly enforce its policies and procedures.
Accessing official examination content prior to testing, breaching the confidentiality of examination content, or any
attempt to subvert the examination process represent violations of test regulations. Conduct occurring before,
during, or after testing that violates the examination regulations and rules of conduct could result in invalidation of
examination results and other penalties.
Candidates must be truthful in completing the application and must abide by all instructions regarding examination
conduct. Failure to comply with the examination regulations and rules of conduct could result in a determination of
the presence of an irregularity, and examination results could consequently be voided. If results are voided as a
result of an irregularity, candidates could be prohibited from testing for a specified time period or, in egregious
cases, from retesting at all. Additionally, candidates could face civil or criminal prosecution.
By applying for the examination, candidates agree to abide by the following Rules of Conduct:
1. The candidate certifies that they are registering for this examination for the purpose indicated in the
Examination Purpose section of this Candidate Guide. The examination may not be taken on behalf of
anyone else or for any reason other than for the purpose indicated. Candidates may not take the
examination to practice or to obtain an advance review of the content
2. Candidates are not allowed to complete an examination for any reason other than that indicated by the
Examination Purpose. If available information suggests a previously eligible candidate might be
completing an examination for other purposes, the Examination Program may revoke the candidate’s
eligibility, and the candidate could be required to re-establish eligibility to take the examination.
3. Candidates will not give, receive, or obtain any form of unauthorized assistance prior to the examination,
during the examination (e.g., in the testing room or when on a break), or subsequent to the examination.
4. Candidates will maintain the confidentiality of examination content at all times. Candidates will not
reproduce or attempt to reproduce examination materials through memorization, recording, copying, or
other means. Candidates will not provide information concerning examination content that might affect the
examination’s ability to accurately assess candidates’ skills, or that might provide unfair advantage to
other candidates. For example, Candidates will neither make use of nor participate in the sharing or
distribution of information regarding examination content or answers (via electronic means or otherwise).
5. Candidates will not bring any unauthorized materials, as listed in the Examination Misconduct section of
this Guide, into the secure testing area. All unauthorized materials must be placed in an assigned locker
and may not be accessed during testing.
6. Candidates will not remove information about the exam (written, printed, recorded, or other) from the test
center.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 24
7. Candidates will comply with Administration Vendor test center policies and procedures and will not create
a disturbance in the test center.
8. Candidates will not tamper with the computer testing equipment and facilities.
9. Candidates will cooperate fully with any investigations involving testing irregularities and agree to have
their examination analyzed to detect aberrancies.
10. This Examination is a secure examination protected by U.S. copyright laws. Any unauthorized disclosure
of the examination’s content could result in civil liability, criminal penalties, voiding of examination results,
or other appropriate penalties.
Test content (in whole or in part) is prohibited from being disclosed before, during, or after the test to anyone,
including but not limited to: family, friends, classmates, colleagues, or test preparation organizations. This applies
to any content disclosed through discussions, emails, in writing, online (e.g., blogs, social media, websites), or
otherwise.
Candidates who receive unreleased test items should immediately contact DTS at testsecurity@ada.org, and
should NOT review the materials they have received. Candidates who have been found to be in possession of
such information—or to have participated in the distribution of this information—may have their examination
results voided. Penalties might be imposed subsequent to discovery and investigation of the original incident,
which could occur years after the incident itself.
Candidates are encouraged to report any activities where information about examination questions is disclosed,
so that DTS can investigate and take any necessary action. Report such activity to DTS at testsecurity@ada.org.
PRIVACY AND SECURITY
The Examination Program will maintain the privacy and security of candidates’ personal information using industry
standard methods. DTS will collect and retain personal information to serve candidate needs, administer the
examination, fulfill Examination Program responsibilities (e.g., to maintain the integrity of the test and detect and
prevent unlawful activity), and fulfill legal requirements. Examination results shall be retained indefinitely, along
with testing records and candidates’ personal information.
Before the examination is administered, candidates will be required to provide a written release concerning the
collection of their biometric information. Biometrics are collected by the Administration Vendor for purposes of
verifying identity and detecting and preventing unlawful activity; the data is stored securely by the Administration
Vendor.
As applicable and in accordance with the purpose of each specific Examination Program, examination results will
be released or reported to state boards, education programs, or other entities upon written candidate
authorization or designation by electronic means through the electronic application or score report request form.
Examination results may be released or reported in the absence of such authorization when policies indicate that
such notification is appropriate (e.g., notification of state boards concerning an irregularity). Examination results
may be released to education programs to enable those programs to understand student outcomes. For research
and policymaking purposes, examination results may be released — with personally identifying information
removed — to individuals or entities that the Examination Program deems legitimately interested. Information
regarding privacy policies is made available to all candidates and the public.
The ADA provides information technology support for the Examination Program and uses data security
procedures to protect the integrity of personal and exam information. Security safeguards include administrative,
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 25
technical, and physical safeguards over data and data processing systems. For information on policies relating
to your use of the ADA website, please refer to the Privacy Notice and Terms of Use available at
ADA.org/privacy-notice.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 26
EXAMINATION MISCONDUCT
The Examination Program strives to report results that accurately reflect the skills and performance of each
candidate. The standards and procedures for administering each examination are intended to provide candidates
with a reasonable opportunity to demonstrate their skills, and to facilitate accurate evaluation of those skills.
The Examination Program reserves the right to withhold, void, or invalidate any result when, in the Examination.
Program’s judgment, it is reasonable to question the validity of the result. Reasons for withholding, voiding, or
invalidating results, or for imposing other appropriate penalties, could include, but are not limited to, the following:
• Unusual answer patterns
• Atypical score increases from one examination attempt to another
• Discrepancy in, or falsification of, a candidate’s identification
• Information indicating that a candidate has engaged in misconduct or a violation of the examination
regulations, rules of conduct, or test center procedures
• Sharing of remembered exam questions or answers. This includes sharing through social media
platforms, online discussion forums, or other means
• Taking an examination on behalf of another individual, or having another individual take an examination
on your behalf
• Falsification of application information or supporting documents
• Falsification of a candidate’s results or results report
• Inconsistent performance on different sections of the exam from one examination attempt to another
• Improper access to secure exam content
• Evidence is available concerning the presence of an examination administration irregularity
• Any other information indicating the results might not be valid.
When examination results are voided or invalidated, the candidate is notified in writing. This notice includes
information about the decision and the procedure for appeal. Results will remain voided until an appeal process
has been completed, or the time for appeal has expired.
When previously reported results are voided, the score report recipient will be notified in writing that the result has
been voided.
If it is determined a candidate has engaged in irregular behavior, information regarding this determination
becomes a part of the candidate’s record. At its sole discretion, the Examination Program may elect to send a
summary report documenting the incident to legitimately interested parties. This would include all parties to whom
a candidate has instructed results be sent (both currently and in the future).
In situations where an irregularity has occurred, individuals who are involved or implicated with respect to the
occurrence of the irregularity, or who are reasonably believed to have witnessed the irregularity, could be asked
to provide information concerning the irregularity.
The Examination Program reserves the right to pursue other remedies, including prosecution of anyone
whose conduct unlawfully undermines the security of the examination or the integrity of the examination
process.
No personal items are permitted in the secure testing area. Any personal belongings brought to the test center
must be stored in a designated locker; storage is limited. Personal belongings may be inspected. Notes or any
materials accessed during the examination or on an unscheduled break could be confiscated. Accessing personal
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 27
belongings or a locker during an unscheduled break violates the examination regulations. Test administrators are
not authorized to provide permission to candidates to access personal belongings or lockers during an
unscheduled break.
Items that are prohibited from the secure testing area include, but are not limited to, the following:
• Books, notes, study materials, scratch paper, tissues, and markers
• Personal earplugs not previously approved by the Administration Vendor. Headphones NOT provided to
you by the Administration Vendor.
• Dental instruments, models, or materials
• Slide rules, paper, calculating devices, rulers, and other measuring devices (except those items approved
in advance under testing accommodations)
• Electronic devices such as cell phones, recording devices, iPods, tablets, and headsets
• Tote bags, purses, wallets, backpacks, and briefcases
• Highlighters, pens, erasers, pencils, dictionaries, and translators
• Food, candy, gum, and beverages (except those items approved in advance under testing
accommodations)
• Outerwear, such as coats, jackets, gloves, or head coverings (religious attire is allowed)
• Good luck charms, statues, religious or spiritual items, and similar objects
• Watches (digital, analog, or smart) or timing devices (a timer is provided on the computer screen during
the examination)
• Magnifying devices
• Jewelry (except for wedding and engagement rings)
The test center will provide two note boards (without graph lines) and two low-odor fine tip markers during the
examination. Used note boards will be replaced by test center staff upon request. Scratch paper, pencils, and
markers not furnished by the testing center are prohibited. You are not guaranteed to receive graph lines on your
provided materials.
Candidates may not write on the note boards before the test begins or during scheduled breaks. The note boards
should not be folded, bent, distorted, or modified in any manner. Markers cannot be used on any surface other
than the note boards. Candidates may not touch the monitor during the examination. All items provided must be
returned to the test administrator before leaving the test center. Test center note boards will not be stored for
multiple day examination use. Any notes taken will be surrendered at the end of each testing day and erased.
Candidates may not engage in conversation with others during testing or while on an unscheduled break.
Discussing the examination is strictly prohibited.
Test center administrators will report the activity of candidates who take unscheduled breaks.
During an unscheduled break, candidates may NOT access personal belongings or prohibited items,
study or refer to notes or texts, use a telephone, eat or drink any food or beverages from lockers, or leave
the test center. Test administrators are NOT authorized to provide permission to engage in these
activities.
Although the examination is administered under strict supervision and security, examination irregularities can
sometimes occur. Examination results could be voided based upon a breach of examination security, invalid
examination conditions, or candidate violations of the examination regulations, rules of conduct, or test center
procedures. If irregularities are detected during the examination, or evidence of irregular behavior is disclosed
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 28
when the examination is scored or afterward, those involved will have their examination results voided and face
appropriate penalties.
Failure to comply with examination regulations, rules of conduct, and test center procedures could result
in a determination of the presence of an irregularity, and examination results could be withheld, canceled,
considered invalid, or another appropriate penalty could be imposed. Candidates might also be directed
to leave the test center before the examination is completed. If results are withheld or invalidated, or
other penalties are proposed or imposed as the result of an irregularity, candidates could be prohibited
from testing and other appropriate penalties could be imposed.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 29
IRREGULARITIES AND APPEALS
An irregularity is defined as a situation in which there could be a question about the validity of examination results
in accurately reflecting the ability and skills of a candidate.
For example, such questions could be raised when:
• there is communication between candidates during the testing session.
• unauthorized assistance occurs.
• candidates have inappropriate access to examination content (e.g. remembered questions or answers
are shared by email, online posting, or other means).
• conduct prohibited by the examination regulations, rules of conduct, or test center procedures occurs or
examination administration disruptions are present, including natural disasters and other emergencies.
When an irregularity is identified, results for the candidate(s) involved will be voided pending resolution of the
corresponding appeal(s). If an appeal is denied or no appeal is filed, the results of the candidate(s) involved could
remain voided and/or other appropriate remedies imposed.
Rule violations and/or irregularities occurring in one Examination Program implemented by DTS may result in
penalties that impact a candidate’s ability to test in another Examination Program implemented by DTS.
Candidates whose results are subject to being voided are notified by written correspondence and provided with a
copy of the Limited Right of Appeal for Examination Candidates. Appeals must be submitted in writing within 30
days of notification of the irregularity. Appeals must be submitted in writing within 30 days of notification of
the irregularity. Appeals must clearly state the specific relief being requested and include corresponding
arguments, evidence, and documentation in support of the request.
The candidate will be notified of the appeal decision within 60 days after receipt of the appeal.
When considering an appeal, the Examination Program strives to ensure that examination results accurately
reflect candidates’ skills, and that the appealing candidate has an opportunity equal to, but not greater than, the
opportunity provided to other candidates.
Results will be voided when there is a reasonable and good faith basis to do so. If it is determined that voiding
results is not warranted under the circumstances, the results will be released.
Candidates should be aware that irregularities — other than natural disasters and emergencies beyond the
control of the candidate — are considered to be a serious breach of the examination process.
The Examination Program strives to handle irregularities and their investigation in a confidential, professional, fair,
and objective manner.
Candidates should be aware that reports of irregularities may have consequences beyond the voiding of results or
the imposition of other appropriate penalties.
• Information regarding the irregularities may be brought to the attention of school authorities, regulatory
agencies, or other entities, by other sources.
• Additional information concerning a candidate may surface within the context of an investigation into an
irregularity.
© 2024 American Dental Association All rights reserved. Updated: 08/22/2024
DAT 2024 Candidate Guide | 30
Candidates are encouraged to report suspicious activity or observations of violations of the examination
regulations to DTS at testsecurity@ada.org.
ARBITRATION REQUIREMENT
Arbitration has become an increasingly common way to resolve legal differences. Generally speaking, the
advantages of arbitration over traditional lawsuits are that arbitration is less expensive and issues are resolved in
less time. If a candidate wishes to pursue a dispute that has not been resolved by the appeal process mentioned
above and detailed in the Limited Right of Appeal for Examination Candidates, the candidate must use the
procedure described in the following Agreement to Arbitrate.
AGREEMENT TO ARBITRATE
1. In the event that any legal dispute arises between you and the Examination Program, in connection with
your participation in this Examination Program, where that dispute is not resolved by the appeals process
detailed in this Guide and elsewhere, you agree that the exclusive means for resolving the dispute shall
be Binding Arbitration as described by the terms of this Agreement. This means that you waive the rights
you may have to resolve the dispute in a court of law, or by any other means that might otherwise be
available to you.
2. The American Dental Association (“ADA”) agrees to be similarly bound except that the ADA reserves its
full rights to pursue injunctive and other appropriate relief in any state or federal court in cases of unfair
competition or violations of, or threats to violate, any intellectual property rights of the ADA. With respect
to any action filed by the ADA pursuant to this paragraph 2, you consent to submit to the jurisdiction of the
state or federal court in which the ADA seeks relief.
3. Arbitration proceedings initiated pursuant to this Agreement to Arbitrate shall be conducted in accordance
with the then current Consumer Arbitration Rules of the American Arbitration Association. The arbitrator’s
award shall be binding and may be entered as a judgment in any court of competent jurisdiction.
Information about the American Arbitration Association, its rules, and its forms are available from the
American Arbitration Association website, www.adr.org.
4. In the event of Arbitration, and except to the extent the Consumer Arbitration Rules provide otherwise, the
parties shall bear their own costs and attorneys’ fees associated with the Arbitration proceedings, unless
the arbitrator directs one of the parties to pay the other’s costs, or attorneys’ fees, or both.
5. To the fullest extent permitted by law, no Arbitration brought pursuant to this Agreement shall be joined to
any Arbitration involving any other party whether through “Class Arbitration” proceedings or otherwise.
6. This Agreement is part of the application to take this examination. Your assent to be bound by it is a
requirement for taking the examination, but you can only sit for the examination if you also fulfill all other
conditions imposed by the Examination Program.
